Fluorescence Cuvette Guide: 4-Sided Polish, Materials & Volume
On This Page
- Why fluorescence needs a different cuvette
- The 6 surfaces of a cuvette
- Polish quality: numbers that matter
- Autofluorescence: the silent killer
- Path length & sample volume
- Open vs masked apertures
- Cap, septum & oxygen quenching
- Fluorometer compatibility
- Sample-prep tips
- Decision tree
- Recommended products
- FAQ
Choose the Right Fluorescence Cuvette: 4-Sided Polish, Low Autofluorescence, Tiny Volumes
🧼 Need to clean these cells? See the Cuvette Cleaning Protocol — 10 cleaning agents, 6 analyte cases, daily & deep cleaning steps.
All four side faces optically polished, sub-2-nm RMS roughness, and a substrate that doesn’t glow back at you — what really matters when you buy a fluorescence cell, and how to avoid the four mistakes that kill your spectrum.
Why fluorescence needs a different cuvette
A UV-Vis absorbance measurement is a one-axis experiment. Light enters the front face of the cell, passes through the sample, and exits the back face into the detector. The two side faces never see optical signal — the manufacturer can leave them frosted to save grinding time, and many absorbance cuvettes ship that way.
Fluorescence detection is a two-axis experiment. The excitation lamp delivers UV or visible photons through one face; the sample absorbs them and re-emits photons in every direction. Because the sample re-emits across the full 4π sphere, the detector is placed perpendicular to the excitation beam — typically the right or left side face — to suppress contamination from the excitation source itself. That right-angle geometry is the entire reason a fluorometer can detect emission against the background of an intense source.
The catch: the perpendicular faces now carry optical signal too. Both the entry and exit faces of the excitation axis must be polished, and the two faces flanking the emission axis must also be polished. Four polished side faces, not two — hence the term four-window or four-sided polish cuvette.
If you slide a 2-window absorbance cuvette into a fluorometer holder, the two frosted side faces act as diffuse scatterers. Some of the excitation beam reflects off the rough surface and bounces directly into the perpendicular detector path. The result is a baseline that rides at 5–20% of the excitation intensity — a noise floor large enough to bury most analyte fluorescence below it. Customers occasionally try this at the request of a budget-conscious manager and call us with a “broken” instrument that turns out to be working perfectly with the wrong cell.
For the same reason, a 4-window cell can substitute for a 2-window absorbance cell with no penalty (you simply don’t use the extra polished faces). When a single sample needs both absorbance and fluorescence — common in nanoparticle, dye-laser, and photophysics work — buying a 4-window cell once is cheaper than buying two cells. See the UV-Vis Spectrophotometry Guide for the absorbance-side rationale.
The 6 surfaces of a cuvette
Every rectangular cuvette has six surfaces: four side faces (front, back, left, right), one top opening, and one bottom. Each face has a different optical role, and each can be left frosted, ground, or finished to optical polish depending on the application.
For absorbance work the front and back faces — the path-length faces — must be polished and parallel; the two side faces and the bottom can stay frosted. For fluorescence, all four side faces must be polished. The top is either left open or fitted with a stopper, septum, or PTFE cap; the bottom is intentionally frosted because the cell holder rests on it and a polished bottom would slip and shift between measurements.
Manufacturers usually identify cuvette types by the count of polished faces:
- Type 1 / Two-Way Light: 2 polished faces (front + back). UV-Vis absorbance only.
- Type 4 / Four-Way Light: 4 polished side faces. UV-Vis absorbance plus fluorescence.
- Type 1F / All-Polished: 4 polished sides + polished top and bottom. Ultraviolet circular dichroism (CD), polarimetry, and certain laser experiments where stray light through the top/bottom matters.
If you only see “Type 4” on a datasheet, that means four polished side faces — the standard fluorescence configuration. MachinedQuartz catalogs these as Four-Way Light in our SKU naming convention.
Polish quality: numbers that matter
“Polished” is not a binary spec. The relevant numbers are surface roughness (RMS), flatness, and parallelism — and these vary by an order of magnitude between premium and budget cells. For fluorescence, the difference shows up in your spectrum as baseline noise.
| Spec | Premium fluorescence cell | Standard absorbance cell | Disposable plastic |
|---|---|---|---|
| Surface roughness (RMS) | ≤ 2 nm | 5–10 nm | 20–100 nm (mold finish) |
| Flatness | λ/4 @ 633 nm | λ/2 | not specified |
| Parallelism | ≤ 30 arcsec | 1–2 arcmin | not specified |
| Transmission @ 250 nm | > 88% | > 80% | ~0% (opaque) |
A rough surface scatters incident light into a wide cone. With 2 nm RMS roughness — premium fused-silica polishing — the scatter cone is so narrow that > 99% of the excitation beam continues forward into the back face and the perpendicular axis sees only the sample’s true emission. At 20 nm RMS, several percent of the excitation photons leak sideways every time the beam crosses a surface. In a 1-cm cuvette the beam crosses two faces (entry and exit), so the cell behaves like a small scatter source even before any sample is added.
Flatness matters for a related reason. A face that bows by λ/2 (≈ 316 nm at 633 nm reference) acts as a weak prism, displacing the beam by tens of micrometers between top and bottom of the cell. For wavelength-selective fluorescence — say, fluorescence anisotropy or polarization measurements — that displacement reads as a signal artifact.
Parallelism matters when you swap cells between samples. If two cells in the same matched set differ in parallelism by more than 1 arcmin, the beam reaches the detector at a slightly different angle and the integrated intensity changes by 1–3%. That’s why matched fluorescence sets are sold with parallelism specified to a tighter tolerance than single cells.
Autofluorescence: the silent killer
Even with a perfectly polished 4-window cell, the substrate itself can emit fluorescence when illuminated by UV. This is called autofluorescence, and it is the single most common reason that a fluorescence experiment looks “noisy” when in fact the noise is coming from the cuvette wall — not the sample.
Autofluorescence comes from trace impurities and structural defects in the substrate. Borosilicate and soda-lime glasses contain trace iron, manganese, and rare-earth contaminants that fluoresce in the 320–500 nm range when excited below 350 nm. Polystyrene and PMMA disposable cells include UV-absorbing additives and stabilizers that fluoresce strongly in the same band. Fused quartz, by contrast, is essentially pure SiO₂ with sub-ppm metal content; under 280 nm excitation a JGS1 or JGS2 cell emits a baseline that is statistically indistinguishable from a dark count.
The takeaway is wavelength-dependent:
- Excitation below 300 nm (tryptophan at 280 nm, tyrosine at 274 nm, NADH at 290 nm): only fused-silica quartz works. JGS1 grade is preferred because its transmission extends down to 170 nm; JGS2 is acceptable above 220 nm.
- Excitation 300–400 nm (NADH at 340 nm, FAD at 380 nm, common BODIPY dyes): quartz is still preferred; high-quality optical glass (BK7) is acceptable for moderate-sensitivity work; borosilicate is risky.
- Excitation above 400 nm (GFP at 488 nm, TAMRA at 555 nm, Cy5 at 633 nm): glass works fine, and disposable polystyrene is usable for screening assays where 5–10% material background is acceptable.
The chart below maps the most common fluorophores onto these wavelength zones. Match the leftmost dot (excitation peak) to the material zone that contains it — that gives you the cuvette material; the rightmost dot (emission peak) tells you the wavelength range your detector needs to see.
The hidden problem with glue: Standard 80 cells in deep UV
Fused-silica cuvettes are themselves not all equal. The fabrication method determines whether the cell has any organic adhesive in the optical path:
- Standard 80 cells are assembled from five precision-ground plates joined with a UV-cure or thermal-cure adhesive at the seams. The adhesive sits a few millimeters from the optical aperture. For visible and near-UV work the glue is invisible — but at 280 nm excitation some adhesive formulations show a faint emission band around 340 nm that bleeds into the tryptophan window. Most labs never notice this because their analyte is far brighter than the artifact, but in trace measurements it shows up as a 1–3% background.
- Sintered 83 cells are built by fusing fine quartz powder into a one-piece body under heat and pressure. There is no adhesive, no organic component, and no thermal-history seam in the optical path. Transmission stays above 83% across 200–2500 nm and the cell tolerates strong solvents (chromic acid, piranha, hot HNO₃) that would destroy a Standard 80 unit.
- Molded 83 cells go one step further: the entire cell is integrally fused from a single quartz preform, so the wall thickness is continuous from face to face. Thermal stability extends to 1200 °C; for fluorescence at deep-UV excitation the molded geometry minimizes any internal scatter from interface boundaries.
If your fluorescence experiment uses below-300-nm excitation and you care about background levels at the 0.5% scale — pharmaceutical QC of protein formulations, single-molecule prep work, or fluorescence anisotropy on small fluorophores — specify Sintered 83 or Molded 83 and skip the Standard 80 product line entirely. For the full fabrication-method comparison see the fabrication method glossary; for material trade-offs against soda-lime and borosilicate glass see quartz vs glass cuvette.
Path length & sample volume
The default 10 × 10 × 45 mm fluorescence cuvette holds about 3.5 mL and provides a 10 mm excitation path. It works for routine quenching, anisotropy, and emission scans of moderately concentrated samples. The default isn’t always the right choice.
Two scenarios call for a different geometry:
Sub-microliter samples
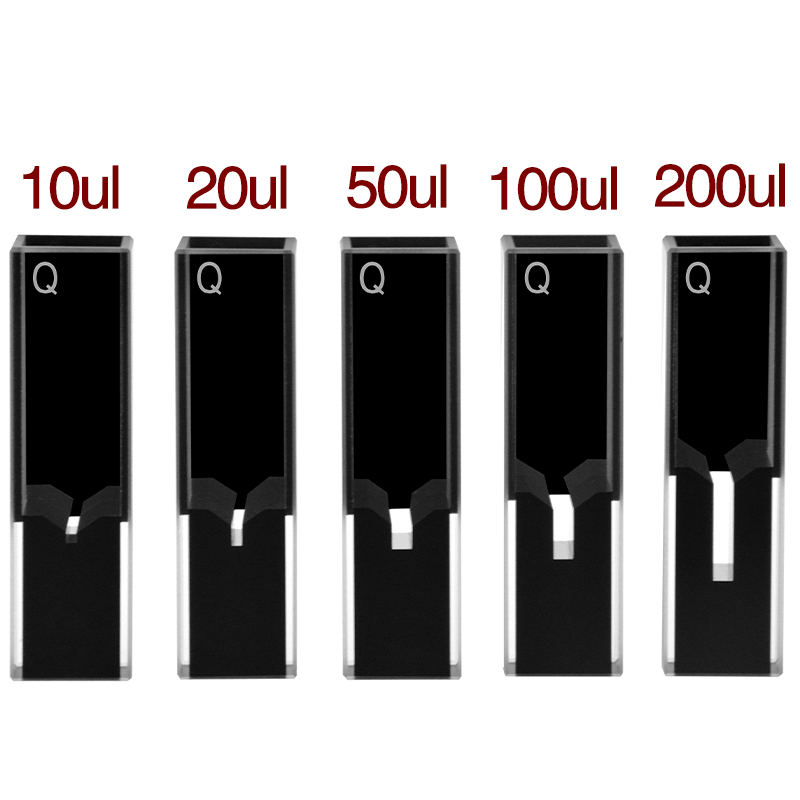
Protein engineering, single-cell extracts, and CRISPR-Cas activity assays often produce only 5–50 µL of sample. A standard 3.5 mL cuvette would dilute that sample by two orders of magnitude before measurement. Sub-micro cells solve this with a small chamber inside a standard 12.5 × 12.5 × 45 mm body — the outer dimensions still fit a fluorometer holder, but the sample volume drops to 5, 10, 20, 50, or 100 µL.
The MachinedQuartz Four-Way Light Ultra-Micro line covers volumes from 5 µL to 200 µL with a 10 mm path. All four side faces are polished so the same cell handles both fluorescence and absorbance. See the Z dimension explanation for how the chamber height aligns with your fluorometer’s optical center — a parameter that catches first-time sub-micro buyers and produces apparently dim spectra when the chamber sits above or below the beam.
Concentrated samples — the inner filter effect
Fluorescence intensity is linear in fluorophore concentration only when the sample’s absorbance at the excitation wavelength stays below about 0.1 in the path used. Above that, two artifacts kick in: the primary inner filter (the front of the cell absorbs the excitation before it reaches the geometric center where the detector is reading) and the secondary inner filter (emitted photons are reabsorbed by ground-state fluorophore on the way out).
The simplest fix is a shorter path. Switching from a 10 mm to a 2 mm path cuvette drops the effective absorbance by 5×, restoring linearity for samples up to 0.5 OD at the excitation wavelength. Below are the path lengths most labs keep on hand:
| Path | Volume (standard chamber) | Use case |
|---|---|---|
| 10 mm | 3.5 mL macro · 1.4 mL semi-micro · 5–200 µL ultra-micro | Default; dilute samples |
| 5 mm | 0.7 mL · 1.5 mL chamber | Moderate concentration |
| 2 mm | 0.35 mL · 0.7 mL chamber | High concentration; inner filter avoidance |
| 1 mm | 0.14 mL · 0.35 mL chamber | Very high concentration; lifetime measurements |
The product line covers four geometric tiers with the same 12.5 × 12.5 × 45 mm outer body — pick the chamber size that matches your sample, not the holder.
For a deeper treatment of path-length physics including the Beer-Lambert tradeoff for absorbance work, see the cuvette path length guide and try the Beer-Lambert path length calculator or the cuvette size calculator for fast volume-to-SKU lookup.
Open vs masked apertures
A standard 4-window cuvette has all four side faces clear from corner to corner. A masked or black-walled cell coats two opposite faces — usually the top and bottom — with opaque black PTFE or black-doped quartz so light can only enter and exit through a defined aperture in the middle.
Black walls do two things. First, they absorb light that would otherwise bounce off the inside surfaces of the top and bottom and reach the detector through the perpendicular axis as stray reflection. In low-concentration fluorescence — single-photon counting, dilute fluorophore titrations, and time-correlated single-photon work — that stray-light contribution can be a major source of dark counts. Second, the masked aperture defines exactly the volume of sample the detector sees, so cell-to-cell variation is reduced when running calibration curves.
The trade-offs are real. Black PTFE is harder to clean than bare polished quartz; certain solvents (DMF, DMSO at elevated temperature, hot piranha) attack the black coating over time. Masked cells cost roughly 2× a standard 4-window cell. For routine fluorescence at sample concentrations above the µM range, an open 4-window cell is sufficient and the extra spend on masked cells is wasted.
Use a masked cell when:
- Your fluorophore concentration is below 100 nM and you can see dark-count variation between empty and sample reads
- You’re doing single-photon counting or photon-correlation spectroscopy
- Your fluorometer has a wide-aperture detector (some Edinburgh and PicoQuant single-photon systems)
- You need defined-volume cells for matched-set calibration in pharmaceutical QC
Use a standard open cell when:
- Concentration is above µM
- You routinely clean with chromic acid, piranha, or hot solvent (the black coating is a wear point)
- You’re at the prototyping stage — open cells are cheap to replace if dropped
Cap, septum & oxygen quenching
Fluorescence is sensitive to dissolved oxygen because O₂ is a triplet-state quencher. The fluorescence intensity of long-lived fluorophores — pyrene, naphthalene, ruthenium tris(bipyridine), most lanthanide complexes — can change by 30–80% between aerated and degassed samples. Even shorter-lived dyes like fluorescein lose 5–15% intensity in air-saturated water versus N₂-purged buffer.
The cuvette cap controls how easily oxygen re-enters the sample after preparation:
| Cap type | Seal quality | Best for |
|---|---|---|
| Open / no cap | None | Quick measurements, no O₂ sensitivity |
| Snap-on PTFE | Loose, finger-tight | Routine work, dust prevention |
| Screw cap with PTFE liner | Tight against bulk evaporation | Long acquisitions, anisotropy, viscosity-tracked samples |
| Septum (rubber or PTFE-faced) | Gas-tight; allows syringe injection | Anaerobic work, N₂/Ar purging, kinetic injections |
| Glass stopper, ground-joint | Gas-tight after greasing | Long-term storage; classical photophysics |
For deep-UV work involving tryptophan lifetime measurements, anaerobic sample preparation is essential — a 5-minute N₂ purge through a septum cell typically extends Trp lifetime from ~2 ns to ~3 ns and recovers up to 40% of intensity. Match the cap material to your buffer chemistry: PTFE works in nearly all aqueous and most organic buffers; silicone is fine for aqueous but swells in DMF and DMSO; viton is needed for prolonged contact with hot acids or oxidizers.
Fluorometer compatibility
Most modern bench fluorometers accept the same standard outer dimension: 12.5 × 12.5 × 45 mm. The internal cell holder positions the optical center about 15 mm from the bottom. As long as your cuvette fits this envelope, it physically slots into nearly every research-grade instrument.
| Instrument | Accepts standard 12.5 × 12.5 × 45 mm | Optical center height (Z) | Notes |
|---|---|---|---|
| Agilent / Varian Cary Eclipse | Yes | 15 mm | Standard four-cell holder; supports all MQ Type 4 cells |
| Horiba FluoroMax-4 / FluoroLog-3 | Yes | 15 mm | FL-1057 holder for sub-micro; specify Z = 15 mm cells |
| Edinburgh FS5 / FLS920 / FLS1000 | Yes | 15 mm | Use masked cells for low-concentration TCSPC work |
| JASCO FP-8000 series (FP-8200 / 8300 / 8500) | Yes | 15 mm | Single-position holder; manual exchange |
| PerkinElmer LS 55 / LS 45 / FL 8500 | Yes | 15 mm | Plate adapter optional for 96-well work |
| Hitachi F-7100 / F-7000 | Yes | 15 mm | Confirm sub-micro Z = 15 mm; some legacy holders use 8.5 mm |
| Thermo Lumina | Yes | 15 mm | Compatible with all Type 4 cells |
| Shimadzu RF-6000 / RF-5301PC | Yes | 15 mm | Solid-sample holder accessory exists |
| Tecan Spark / Spark 10M | 96-well + cuvette | 15 mm cuvette mode | Use cuvette mode for sensitive single-sample work |
| BMG CLARIOstar / PHERAstar | Plate-based; cuvette adapter optional | — | Cuvette adapter takes Type 4 cells |
Two specs to confirm before ordering:
- Outer dimensions: standard is 12.5 × 12.5 × 45 mm (W × D × H). A small number of legacy or specialty fluorometers use 10 × 10 × 45 mm or 12.5 × 12.5 × 48 mm — check your manual.
- Z dimension (chamber center height above bottom): 15 mm is the modern standard. Older Hitachi and some Shimadzu holders used 8.5 mm — a sub-micro cell with the wrong Z will sit above or below the beam, producing apparently dim spectra that are actually optical alignment issues.
MachinedQuartz Type 4 cells ship with the modern 15 mm Z by default. For older instruments, custom Z dimensions are available with 5–7 day lead time — see the full cuvettes & cells size chart for outer-dimension and Z-dimension options, or contact us for a custom quote.
How MachinedQuartz compares to Hellma, Starna and FireflySci
Hellma, Starna and FireflySci are the three legacy brands most labs see on existing inventory. The optical specs of premium 4-window cells are nearly identical across all four manufacturers — the differences are in price, lead time, and fabrication transparency. Below is a direct spec-by-spec comparison for an equivalent 10 mm 3.5 mL macro Four-Way fluorescence cell:
| Spec | Hellma type 101.015 | Starna type 23 | FireflySci Type 4 | MachinedQuartz Four-Way |
|---|---|---|---|---|
| Material | Quartz Suprasil | Spectrosil quartz | UV/IR quartz | JGS1/JGS2 quartz |
| Polish (RMS) | ≤ 2 nm | ≤ 2 nm | ≤ 2 nm | ≤ 2 nm |
| Transmission @ 250 nm | > 88% | > 88% | > 88% | > 88% |
| Outer dimensions | 12.5 × 12.5 × 45 mm | 12.5 × 12.5 × 45 mm | 12.5 × 12.5 × 45 mm | 12.5 × 12.5 × 45 mm |
| Fabrication options published | One default | One default | Two grades | Three (Standard 80 / Sintered 83 / Molded 83) |
| Lead time (in stock) | 2–4 weeks | 2–6 weeks | 1–2 weeks | 1–3 days |
| Lead time (custom) | 8–14 weeks | 10–16 weeks | 4–6 weeks | 4 weeks |
| Single-cell list price (USD) | $280–$420 | $240–$380 | $140–$220 | $80–$150 |
For SKU-by-SKU cross-references see the dedicated comparison guides: Hellma cuvette alternative, Starna cuvette alternative, and FireflySci cuvette alternative, and Azzota cuvette alternative. Each page maps individual part numbers to MQ equivalents with optical specs side-by-side.
Sample-prep tips specific to fluorescence
Once the cuvette is right, the next source of background is the sample preparation itself. Three issues catch most labs:
Particles and Mie scattering
Sub-micron particles scatter light in a cone that overlaps the emission window. Even a clean-looking buffer often has 10⁵–10⁶ particles per mL from filter membranes, cap liners, and tubing. Filter every fluorescence sample through a 0.22 µm PTFE or PES syringe filter before measurement; centrifuge protein samples at 14,000 g for 5 minutes if you can’t filter. The first 100 µL through a syringe filter is usually contaminated by membrane wetting agents — discard it.
Dissolved oxygen
For long-lived fluorophores (microsecond and longer lifetimes — lanthanide chelates, ruthenium complexes, native protein lifetimes near 10 ns and above), purge the buffer with N₂ or Ar for 5–10 minutes before adding the analyte. Use a septum-cap cuvette and inject the sample by syringe to keep the headspace anaerobic. For routine emission scans of nanosecond-lived dyes (fluorescein, rhodamine, GFP) the oxygen effect is small enough to ignore.
Raman water bands
Water has weak but unmistakable Raman scattering bands that appear at fixed frequency offsets from the excitation wavelength — about 3,400 cm⁻¹ for the O-H stretch. With a 350 nm excitation, that produces a peak near 397 nm; with 280 nm excitation it lands near 311 nm. The bands are narrow (~10 nm FWHM) and shift with excitation wavelength, distinguishing them from real fluorescence. Recognize them, don’t fight them — the simplest mitigation is to subtract a buffer-only blank acquired in the same cell at the same wavelength.
Cell cleaning
Fluorescence cells should be rinsed with the sample’s solvent first (to dilute the analyte without precipitating it), then washed with a 1:1 mix of detergent and warm water (Hellmanex III or comparable), then triple-rinsed with deionized water, then ethanol. Allow to air-dry rim-up; do not touch the optical faces. Once a year, soak in chromic acid or Nochromix overnight to remove adsorbed organics — but only if the cell is Sintered 83 or Molded 83. Standard 80 cells degrade in chromic acid because the seam adhesive is attacked.
Decision tree
Use the chart below to narrow down to a single SKU in under a minute. Start with excitation wavelength (top), continue to volume (middle), then check the special requirements at the bottom.
If your decision lands in the < 300 nm × < 200 µL × anaerobic corner — pharma QC of recombinant proteins, Trp lifetime measurements, or native fluorescence kinetics — the spec is a Sintered 83 or Molded 83 Ultra-Micro Four-Way cell with a septum cap. Most labs settle on the 50 µL or 100 µL volume.
Recommended MachinedQuartz products
The Four-Way Light line is built specifically for fluorescence. All cells use JGS1 or JGS2 fused silica, all four side faces are polished to ≤ 2 nm RMS, and outer dimensions are 12.5 × 12.5 × 45 mm with a 15 mm optical-center Z dimension — the modern fluorometer standard.

50 µL Ultra-Micro 4-Way
10 mm path · Z = 15 mm · PTFE cap
View C104CD15 →

100 µL Ultra-Micro 4-Way
10 mm path · Z = 15 mm · PTFE cap
View C104CD16 →

200 µL Ultra-Micro 4-Way
10 mm path · Z = 15 mm · PTFE cap
View C104CD12 →

Macro 4-Way · 3.5 mL
10 mm path · routine fluorescence
Browse macro options →

4-Way + Septum/Stopper
anaerobic · O₂ quenching control
View septum cells →

Custom Sintered 83 / Molded 83
glue-free · 200–2500 nm · 4-week lead
Get a custom quote →
If the right configuration isn’t on this page, see the custom quartz cuvettes page for non-standard path lengths, integrated stopcocks, jacketed (temperature-controlled) cells, and OEM volumes. Lead time on a fully custom Four-Way cell is typically 4 weeks; matched sets ship together with parallelism specified to ≤ 30 arcsec. For a side-by-side comparison of every MQ cuvette family — fluorescence, absorbance, sub-micro, flow-through, and OEM — see the comparative analysis of quartz cuvette models.
Frequently asked questions
No. The two side faces of an absorbance cuvette are usually frosted, and they will scatter excitation light directly into the perpendicular detector path. The result is a noise floor of 5–20% of the excitation intensity that buries most analyte fluorescence. You need a 4-window cell with all four side faces optically polished.
It means all four side faces of the rectangular cuvette body are polished to optical quality, not just the two faces parallel to the excitation path. The top is normally an opening (with or without a cap) and the bottom stays frosted because it sits in the cell holder. The terminology comes from how light “sees” the cell — four optical windows, four polished faces.
Yes, dramatically — especially for excitation below 400 nm. Polystyrene and PMMA disposable cuvettes have heavy autofluorescence across the UV. Borosilicate glass adds a 320–360 nm hump. Optical glass (BK7) is better but still emits below 350 nm. Fused-silica quartz (JGS1 or JGS2) holds a near-zero baseline from 200 to 800 nm. For below-300 nm excitation, only quartz is usable.
An Ultra-Micro Four-Way cuvette with a 50 µL chamber and 10 mm path. The outer body is the standard 12.5 × 12.5 × 45 mm so it fits any modern fluorometer; the small internal chamber holds 50 µL while keeping the same 15 mm Z-dimension as full-size cells. MachinedQuartz Four-Way Ultra-Micro cells cover 5 µL to 200 µL.
Rinse with the sample solvent first, then 1:1 detergent (Hellmanex III or equivalent) and warm water, then triple-rinse with deionized water, then ethanol. Air-dry rim-up and don’t touch the optical faces. For deep cleaning of Sintered 83 or Molded 83 cells, an overnight chromic acid soak removes adsorbed organics — but never use chromic acid or piranha on Standard 80 cells, because the seam adhesive degrades.
Yes for excitation above 450 nm with bright fluorophores (GFP, fluorescein at moderate concentration, phycoerythrin) where 5–10% material background is tolerable. No for native protein fluorescence, NADH, FAD, or any UV excitation — plastic autofluorescence will dominate the spectrum. For trace measurements at any wavelength, use quartz.
Three common causes: (1) cuvette autofluorescence is being washed away as you re-fill the cell — switch to quartz; (2) the cells in your matched set differ in parallelism — confirm spec ≤ 30 arcsec; (3) sub-micron particles are settling in the beam path — filter samples through 0.22 µm before measurement. If all three are addressed and drift persists, check for fluorophore adsorption to the cell wall, especially for cationic dyes on hydroxylated quartz surfaces.
Hellma, Starna, and FireflySci sell cuvettes, not fluorometers — and their cuvettes share the same 12.5 × 12.5 × 45 mm outer dimensions as MQ cells. Our Four-Way cells are direct geometric replacements for Hellma type 101.015, Starna type 23, FireflySci Type 4, and equivalent. See our Hellma alternative, Starna alternative, and FireflySci alternative cross-reference pages for SKU-by-SKU mapping.
5 µL with a Sub-Micro Four-Way cell. Below that, sample evaporation during measurement and meniscus effects start to dominate the signal — consider a fluorescence-capable microplate or capillary cell instead. For 5–200 µL, the Ultra-Micro Four-Way line preserves spectral fidelity at the same level as a full 3.5 mL macro cell.
When concentration is below 100 nM, when you’re doing single-photon counting or photon-correlation spectroscopy, or when matched-set cell-to-cell variation must be below 1%. For routine emission scans at µM and above, an open 4-window cell delivers the same data at half the cost and with easier cleaning.
If your excitation is below 400 nm, yes — full stop. Disposable polystyrene cells run about $0.50 each but contribute 5–20% material background to any UV-excited spectrum. Borosilicate glass cells run $20–40 and contribute ~5% background below 350 nm. A premium fused-silica quartz cell with 4-sided polish runs $80–250 depending on volume and fabrication, but it contributes essentially zero autofluorescence and lasts indefinitely with proper care. For above-400-nm visible-fluorophore work, the cost-benefit shifts toward optical glass; for tryptophan, NAD(P)H, FAD, or any deep-UV work, quartz is the only path to meaningful data.
A standard 4-sided macro 10 mm fluorescence cuvette in JGS2 quartz with PTFE cap runs $80–150 from MachinedQuartz, depending on fabrication method (Standard 80 cheapest, Sintered 83 or Molded 83 for premium UV work). Sub-micro and ultra-micro cells cost $150–250 because the chamber must be precisely machined. A masked black-walled cell adds roughly 2× to the base price. Disposable polystyrene 4-clear-side cuvettes run $0.50 each in pack-of-100 quantities but are limited to above-400-nm work. Hellma and Starna premium cells typically cost 2–4× the equivalent MQ price; the optical specs are often identical.
JGS1, JGS2, and JGS3 are Chinese national standard grades of fused silica that correspond closely to international optical grades. JGS1 is the highest-purity UV grade with transmission down to 170 nm — required for deep-UV fluorescence excitation of phenylalanine (258 nm), tyrosine (274 nm), and any sub-280-nm absorption. JGS2 is general UV-Vis grade transmitting down to 220 nm, the workhorse for most fluorescence labs (tryptophan and longer). JGS3 is IR-grade with high OH content; it transmits well into the IR but absorbs in the UV — not used for fluorescence cells. Specify JGS1 for native protein and far-UV CD work; JGS2 is sufficient for everything else.
Yes, if you specify a 4-window (4-sided polished) cell. A 4-window cell satisfies absorbance requirements (the path-length faces are polished and parallel) and adds fluorescence capability (the perpendicular faces are also polished). The reverse is not true — a 2-window absorbance cell cannot do fluorescence because the side faces scatter excitation light into the perpendicular detector. For labs running both UV-Vis and fluorescence on the same samples (typical in nanoparticle, dye, and photophysics work), buying 4-window cells from the start saves money — you avoid maintaining two separate cuvette inventories.
Glossary of fluorescence cuvette terms
- Stokes shift
- The wavelength difference between an analyte’s excitation and emission peaks. Larger shifts (e.g., NADH 340→460 nm = 120 nm) make 90° fluorescence detection easier; smaller shifts (e.g., GFP 488→507 nm = 19 nm) require sharper monochromators to separate ex from em.
- RMS surface roughness
- Root-mean-square deviation of a polished surface from perfect flatness, measured in nanometers. Premium fluorescence cuvettes spec ≤ 2 nm RMS; budget cells run 5–20 nm. Lower RMS means less stray scatter into the perpendicular detector axis.
- λ/4 flatness
- A surface flat to one-quarter of a wavelength of light, conventionally measured at 633 nm (red HeNe laser). λ/4 = ±158 nm peak-to-valley deviation. Typical premium cuvette spec.
- Quantum yield (Φ)
- Ratio of photons emitted to photons absorbed by a fluorophore. Φ = 1 means every absorbed photon produces one emitted photon (theoretical maximum). High-Φ fluorophores (fluorescein Φ=0.92) tolerate dirtier cells; low-Φ analytes (Trp Φ=0.13 in protein context) demand premium quartz to keep S/N usable.
- Inner filter effect
- Self-absorption artifact in concentrated samples: the front of the cell absorbs most excitation photons before they reach the detector’s geometric center, and emitted photons are reabsorbed on their way out. Signal becomes non-linear above OD ≈ 0.1 at the excitation wavelength.
- Z dimension
- Vertical distance from the bottom of the cuvette to the optical center of the chamber. Modern fluorometers (Cary Eclipse, FluoroMax-4, FS5) use Z = 15 mm; legacy Hitachi and Shimadzu instruments may use Z = 8.5 mm. Mismatch produces apparently dim spectra.
- Autofluorescence
- Background fluorescence from the cuvette substrate itself. Significant for borosilicate glass and disposable polystyrene; near-zero for fused-silica quartz across 200–800 nm. Determined by trace metal impurities, rare-earth contaminants, and (in glued cuvettes) the seam adhesive.
References & further reading
For deeper treatment of fluorescence physics, instrumentation, and assay design, the standard reference is Joseph R. Lakowicz, Principles of Fluorescence Spectroscopy, 3rd edition (Springer, 2006) — see chapters 1–3 for excitation/emission fundamentals and chapter 22 for cell geometry and inner-filter corrections. The IUPAC Compendium of Chemical Terminology (the Gold Book) provides authoritative definitions for fluorescence quantum yield, Stokes shift, and quenching. For neighboring formats — capillary fluorescence flow cells, Raman cells, and fiber-optic-coupled probes — see fused silica vs quartz capillary tubes.
Next steps
Picking the right fluorescence cuvette comes down to four questions: what wavelength, how much sample, how concentrated, and any special requirements. Work through the decision tree above and you’ll arrive at one of three or four SKUs in MachinedQuartz’s catalog — most labs order the Macro 4-Way for routine work and add an Ultra-Micro 4-Way for valuable low-volume samples.
If your geometry is non-standard — a custom path length, a temperature-jacketed body, an integrated stopcock, or a non-standard outer envelope to fit a legacy fluorometer — we build to spec with 4-week lead time. Submit a custom request with your fluorometer model, sample volume, and target wavelength range; you’ll have a quote in 24 hours.






Recent Comments