Micro Cuvette Guide: Semi-Micro, Sub-Micro & Ultra-Micro Selection
On This Page
Pick the Right Cuvette for Tiny Samples
Semi-micro, sub-micro, ultra-micro — when each one is right, the path-length tradeoff, and how the Z dimension determines whether your spectrophotometer sees any signal at all.
The four cuvette volume classes
“Micro cuvette” is an umbrella term covering everything below the standard 3.5 mL macro cell. Three sub-classes sit underneath, each addressing a different problem — and each requires a different chamber geometry, different aperture mask, and different pipetting protocol.
Macro (3.5 mL · 10 × 10 mm path)
The default cuvette. 10 × 10 mm internal cross-section, 3.5 mL working volume, 10 mm optical path. Most published protocols and absorbance-based assays assume this geometry. If you have plenty of sample and don’t care about reagent cost, the macro cell is always the right answer because it’s the most reproducible and most forgiving on pipetting.
Semi-Micro (700–1750 µL · 10 × 4 mm path)
The “save money on reagents” cell. The chamber is the same 10 mm length but only 4 mm wide; volume drops to about 1.4 mL while keeping the 10 mm optical path. Used routinely in labs running large batches of dilute samples — calibration curves, screening assays, environmental samples — where each measurement uses 100 mg of expensive reagent in a macro cell, but only 30 mg in a semi-micro.
Semi-micro and macro share the same fluorometer holder. Switching between them mid-experiment doesn’t require recalibrating Z dimension or beam alignment.
Sub-Micro (typically 200 µL · 10 × 2 mm path or 10 mm masked)
The transition zone. Sample volumes between 100 and 700 µL fall here — too small for semi-micro, too large for ultra-micro. Sub-micro cells use a 10 × 2 mm internal chamber or, equivalently, a masked aperture in a wider body. The 10 mm optical path is preserved through the masked geometry.
Ultra-Micro (5–200 µL · 10 mm masked path)
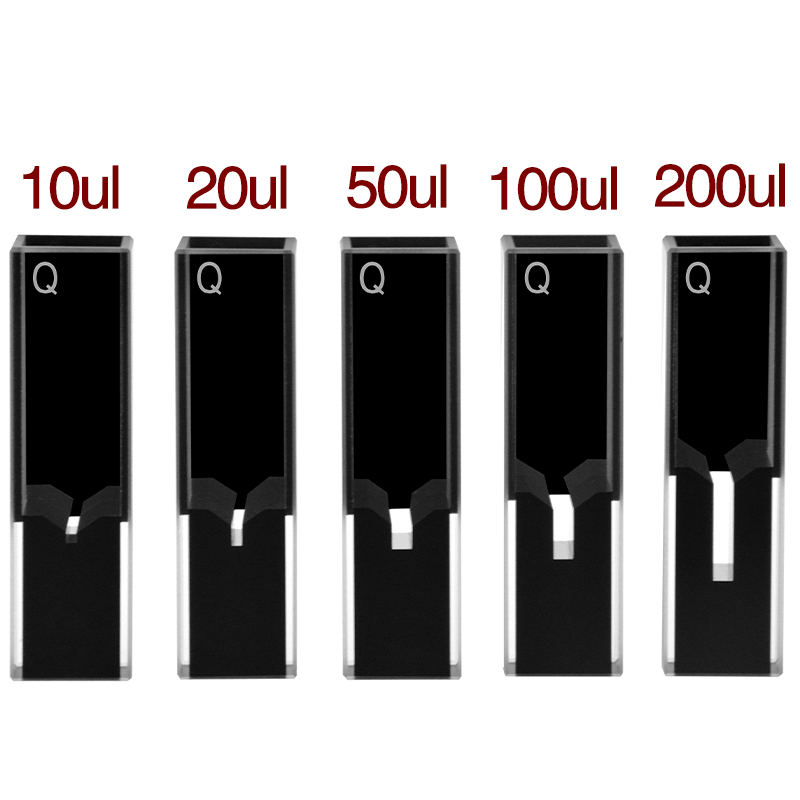
The trace-volume class. The optical path stays 10 mm but the chamber is reduced to a tiny well in the middle of the cell, with opaque masks above and below. Ultra-micro cells cover 5, 10, 20, 50, 100, and 200 µL volumes; the 50 µL is the most-ordered SKU because it’s the practical lower bound for routine pipetting (5 µL pipettors require near-perfect technique to repeat ±5% volume).
Why the same 10 mm path across all four classes? Because most published assays, every fluorometer’s calibration, and the Beer-Lambert constants in standard reference tables assume 10 mm. Changing to 2 mm or 5 mm path shifts your concentration math by exact integer factors but breaks comparison with literature data. For deep concentration work where the 10 mm path saturates, see the cuvette path length guide.
Decision matrix — which class for which sample
The decision tree below collapses to one of five outcomes per sample volume. Most labs settle into a 2-class inventory: a macro for routine work plus an ultra-micro 50 µL for valuable low-volume samples. Heavy fluorescence labs add a Type 4 (four-way light) version of the ultra-micro for trace fluorometric titrations.
Three rules of thumb that catch most edge cases:
- Always order one cell larger than your minimum. A 50 µL pipettor is reliable for samples of 70 µL minimum (with a slight reservoir to avoid air); a 200 µL Ultra-Micro is the safest below 250 µL. Going right to the limit of the chamber means the air-bubble reading you’ll see when the cell is overfilled by 1 µL.
- Match cells to instruments, not the other way. Sub-100-µL chambers usually require Z = 15 mm matched to the instrument; some legacy fluorometers need Z = 8.5 mm and a different chamber depth. Confirm your instrument’s Z value before ordering — the Z dimension lookup tool covers 50+ models.
- For matched sets, buy one extra. Lost or dropped cells in a matched set make the remaining cells worthless for calibration curves. A 5-cell matched set with one spare costs $50 more and saves $400 worth of recalibration when the inevitable accident happens.
Path length × volume tradeoff
For a fixed chamber width, path length and volume are linked: doubling the path doubles the volume. The tradeoff matters when you optimize for sensitivity (path = signal) versus sample availability (volume = constraint).
| Class | Inner geometry | Path | Volume | Best for |
|---|---|---|---|---|
| Macro | 10 × 10 mm | 10 mm | 3.5 mL (3 mL fill) | Routine UV-Vis, fluorescence emission scans |
| Semi-Micro 1.4 mL | 10 × 4 mm | 10 mm | 1.2–1.4 mL fill | Save reagent, calibration curves |
| Semi-Micro 700 µL | 5 × 5 mm | 5 mm | 700 µL fill | High-concentration samples (avoid inner-filter) |
| Sub-Micro 350 µL | 5 × 2 mm | 5 mm | 350 µL fill | Lifetime measurements, anisotropy |
| Ultra-Micro 200 µL | 10 mm masked | 10 mm | 200 µL fill | Sub-mg/mL trace fluorescence |
| Ultra-Micro 50 µL ★ | 10 mm masked | 10 mm | 50 µL fill | Pharma QC, single-cell, CRISPR — most-ordered |
| Ultra-Micro 5–20 µL | 10 mm masked | 10 mm | 5–20 µL fill | FFPE single-extract, ultra-trace |
Two paths most labs miss:
- 2 mm path semi-micro — for high-concentration spectroscopy work (DNA quantitation above 50 µg/mL, antibody preparations above 5 mg/mL), the 2 mm path semi-micro keeps total absorbance below 1.0 OD. The 10 mm path equivalent saturates the detector and produces non-linear readings; the 2 mm path is linear up to 0.5 mg/mL DNA without dilution.
- 40 mm long path — at the other extreme, dilute environmental samples and trace-metal solutions need maximum path. A 40 mm cell increases the readable signal 4× compared to a standard 10 mm cell. Standard fluorometer holders don’t fit 40 mm cells, but most UV-Vis spectrophotometers have a long-path adapter accessory.
Inside chamber geometry — open vs masked aperture
From the outside, all four cuvette classes look identical — the same 12.5 × 12.5 × 45 mm fused-silica body. Inside, the chamber design splits into two fundamentally different approaches, and which approach you have determines how much your instrument’s optical alignment matters.
📡 Working in NIR or IR? See the Quartz IR Cuvette Guide — 190–2500 nm range, water-band path length tips, and fabrication choice.
Open chamber (semi-micro and standard sub-micro)
The internal chamber is a simple rectangular volume of fixed cross-section. Sample fills the chamber from rim to bottom; the optical beam passes through the path-length faces and sees sample anywhere along the chamber height. This is the geometry of every macro cuvette and most semi-micro cells.
The key advantage is mechanical tolerance: a slight Z-dimension mismatch (1–2 mm off) still produces a usable spectrum because the beam can find sample to traverse. Every cell tolerates routine pipetting variability.
Masked aperture (ultra-micro and some sub-micro)
To preserve the 10 mm optical path while reducing sample volume, ultra-micro cells use opaque masks (typically black PTFE or black-doped quartz) that cover the top and bottom of the cell, leaving only a small precision-machined aperture in the middle. The aperture is sized to match the volume — 5 µL needs a 1 mm aperture, 200 µL needs a 4 mm aperture, etc.
This trades mechanical tolerance for sample efficiency. A masked cell with the wrong Z dimension is effectively useless: the optical beam hits opaque mask, transmission goes to zero, and the instrument shows no signal. Specifying the Z dimension correctly is non-negotiable for ultra-micro cells.
Z dimension matters more than anything else
For sub-micro and ultra-micro cells with masked apertures, the single most important specification — more important than path length, volume, or material — is the Z dimension. Get the Z wrong and your $200 cuvette behaves like an opaque tube.
The Z dimension is the height from the bottom of the cuvette to the optical center of the chamber. The two industry standards are:
- Z = 15 mm — the modern standard. Used by virtually every fluorometer made after 2005 (Cary Eclipse, Horiba FluoroMax, Edinburgh FS5, JASCO FP-8000, PerkinElmer LS 55) and most modern UV-Vis spectrophotometers (Agilent, Shimadzu UV-1900, Thermo Evolution).
- Z = 8.5 mm — the legacy standard, kept alive by older Hitachi, some Shimadzu UV-1700 series, and OEM equipment with custom optical paths.
Open-chamber cells (macro and semi-micro) tolerate moderate Z mismatch — the beam can find sample anywhere in the chamber. Masked cells (sub-micro and ultra-micro) cannot tolerate any mismatch — the beam either hits the aperture or it doesn’t.
Three steps to confirm Z compatibility before ordering:
- Check your instrument’s manual for “cuvette holder dimension” or “optical center height”
- If unclear, run the Z-Dimension Lookup Tool (link below) — it covers 50+ instruments with the correct Z value pre-confirmed
- If your instrument is older or non-standard, contact MachinedQuartz with the model number; we’ll cross-reference and quote a custom-Z cell if needed (4-week lead time)
📐 Z Dimension Lookup Tool
Look up your instrument’s Z dimension in seconds. 50+ models pre-cataloged · adapter spacer guide · custom-Z order path.
Open the Z Dimension Tool →Pipetting accuracy in micro-volume work
Once you’re working with sub-100-µL samples, pipetting itself becomes the dominant source of measurement error. The cuvette can be perfect — premium quartz, 2 nm RMS polish, Z-matched to the instrument — and the spectrum will still drift 5% sample-to-sample if the pipetting CV is 5%.
Three practical rules for sub-100-µL pipetting:
Use the smallest pipettor that reaches the volume
A 100 µL air-displacement pipettor pipetting 10 µL has 5–7% CV. A 0.5–10 µL pipettor at the same 10 µL has < 1% CV. Pipettor accuracy is best at the upper end of its range; running near the lower end produces the worst error.
Pre-rinse the tip with the sample
For high-viscosity samples (glycerol mixes, concentrated proteins, DMSO solutions) pre-rinse the tip three times by pipetting the sample in and out before the actual measurement transfer. Without pre-rinse, the first transfer leaves residue inside the tip wall and delivers 3–8% less than the dial reading.
Pipette directly into the cuvette, not through a transfer step
Each transfer step adds a CV term. Direct pipetting into a 50 µL ultra-micro chamber gives single-CV accuracy; transferring through a microcentrifuge tube first doubles the CV.
For samples below 20 µL, consider a positive-displacement pipettor (Microman, Gilson MICROMAN E) which displaces sample with a piston rather than air. Air-displacement pipettors give erratic results below 5 µL because the air cushion compresses unevenly with small samples.
Evaporation, meniscus, and surface tension
Below 200 µL, three physical effects start dominating that don’t show up in macro work:
Evaporation
A 50 µL aqueous sample in an open ultra-micro chamber loses about 0.5 µL per minute at 22 °C and 50% relative humidity — 1% per minute. Across a 5-minute kinetic experiment that’s 5% concentration drift. Workarounds:
- Use a capped cell whenever possible (PTFE snap cap or screw cap)
- For uncapped work, run the kinetics fast and time-stamp every reading
- For long acquisitions in air, pre-saturate the headspace by including a small water reservoir in the cell holder
- For DMSO and other low-vapor-pressure solvents, evaporation is negligible
Meniscus and beam profile
A 5 µL droplet in a 1-mm aperture forms a meniscus that bends light. The optical beam — typically 2–3 mm diameter in a fluorometer — sees both the cell wall (which transmits) and the meniscus boundary (which deflects). The result is a characteristic “double peak” in spectra near the absorbance maximum, caused by partial-beam shadowing.
Fix: ensure the chamber is filled to spec. The MQ Ultra-Micro 5 µL cell is rated to fill exactly to 5 µL — under-filling produces meniscus artifacts; over-filling is impossible because the chamber is precisely volume-calibrated.
Surface tension and bubble formation
Pipetting fast into a small chamber creates micro-bubbles that ruin the spectrum. The bubble adds a refractive index discontinuity and reads as a phantom peak. Slow pipetting (1–2 seconds for the full transfer) and tipping the cell at 30° during fill prevents bubble entrainment.
Cleaning sub-microliter chambers
Cleaning a 5 µL chamber requires a different approach than cleaning a 3.5 mL macro cell. Standard 5 mL Hellmanex pour-and-soak doesn’t work — the chamber is too small to flow detergent through, and capillary forces hold residue against the wall.
For sub-100-µL chambers:
- Use a 5–10 µL pipettor to flush the chamber with 0.5% Hellmanex solution 5–10 times (load detergent, expel, repeat)
- Switch to fresh DI water and flush 10 times
- Submerge the entire cell in Hellmanex bath for the deeper soak (the bath surrounds the cell on all sides; the chamber gradually clears via diffusion)
- Sonicate at 40 kHz for 2 minutes while submerged
- Triple DI rinse, then air-dry rim-up overnight (skip oven drying — concentrates residue at chamber bottom)
For the full multi-analyte cleaning protocol — including chromic acid and piranha guidelines for stubborn residue — see the cuvette cleaning protocol.
Recommended MachinedQuartz micro cuvettes
The Four-Way Light Ultra-Micro line covers 5 µL to 200 µL; semi-micro and macro options are available in matching JGS1 / JGS2 quartz fabrication. All cells share 12.5 × 12.5 × 45 mm outer body and Z = 15 mm chamber center (custom Z = 8.5 mm and other dimensions on request, 4-week lead).

50 µL Ultra-Micro 4-Way
most-ordered SKU · Z = 15 mm
View C104CD15 →

100 µL Ultra-Micro 4-Way
moderate-volume titrations
View C104CD16 →

200 µL Ultra-Micro 4-Way
upper sub-micro range
View C104CD12 →

Semi-Micro 350–1750 µL
5×5 mm to 10×4 mm path
Browse semi-micro →
Ultra-Micro 10–200 µL
10 mm masked path
Browse ultra-micro →

Custom Z / Custom Volume
Z = 8.5 mm legacy · 4-week lead
Get a custom quote →
For path-length and concentration calculations before ordering, use the Beer-Lambert path length calculator and the cuvette size calculator. For full SKU range with filter by Z dimension, fabrication, and cap type, see the cuvettes & cells size chart.
Frequently asked questions
The terms refer to sample volume ranges. Macro cells hold 3.5 mL with 10 × 10 mm internal cross-section. Semi-micro cells hold 700–1750 µL with a narrower 10 × 4 mm chamber. Sub-micro cells handle 200–700 µL, often with a 10 × 2 mm or masked aperture. Ultra-micro cells cover 5–200 µL with a precisely masked aperture in an otherwise standard outer body. All four classes typically share the same 12.5 × 12.5 × 45 mm outer dimensions so they fit any modern fluorometer or spectrophotometer.
5 µL with a sub-µL Ultra-Micro cell. Below that, evaporation, meniscus effects, and pipetting error dominate the measurement; consider a fluorescence-capable microplate, a NanoDrop-style microvolume instrument, or a capillary cell instead. The practical sweet spot for repeatable cuvette measurements is 50 µL — pipetting CV stays under 1%, and the chamber tolerates routine handling.
No, as long as the outer body is 12.5 × 12.5 × 45 mm — the standard fluorometer/spectrophotometer envelope. The masked aperture inside the cell sits at the standard Z = 15 mm height, matched to your instrument’s beam path. The only special accessory you might need is a stir bar or temperature-control jacket; most ultra-micro work uses the standard cell holder unchanged.
Check your instrument’s manual under “cuvette holder” or “optical center height.” Modern instruments (post-2005) almost always use Z = 15 mm. Legacy Hitachi, some Shimadzu UV-1700 series, and older OEM equipment use Z = 8.5 mm. The MachinedQuartz Z Dimension Lookup Tool covers 50+ instruments — see https://machinedquartz.com/z-dimension-of-sub-micro-cuvettes/. If the wrong Z is used, masked-aperture cells produce near-zero transmission because the beam hits opaque mask instead of the sample window.
Yes, with thorough cleaning between samples. Sub-100-µL chambers retain residue more easily than macro cells because of capillary forces. Use 5–10 fill-and-flush cycles with a 0.5% Hellmanex solution, sonicate while submerged for 2 minutes, then triple DI rinse and air-dry. For trace fluorescence work below 100 nM concentration, dedicate a cell to each project rather than risk carryover.
10 mm — the same as a standard macro cell. Ultra-micro cells preserve the 10 mm path length by masking the chamber to a small central aperture; the optical beam still traverses the full 10 mm of sample. This is what makes ultra-micro cells directly comparable to macro published assays — all the molar absorptivity constants in standard reference tables assume 10 mm path.
Yes, when specified as Type 4 / Four-Way Light fabrication with all four side faces polished. Open-chamber semi-micro cells work in any fluorometer; masked-aperture ultra-micro cells need precise Z dimension match to the instrument. For deep-UV fluorescence (tryptophan, tyrosine) specify Sintered 83 fabrication so no adhesive sits in the optical path. Full guide at https://machinedquartz.com/fluorescence-cuvette-guide/.
Use a 5–10 µL pipettor to flush 0.5% Hellmanex through the chamber 5–10 times, then DI water 10 times. Submerge the cell in a Hellmanex bath for 30 minutes, sonicate at 40 kHz for 2 minutes while submerged, then triple DI rinse and air-dry rim-up overnight. Don’t use the same protocol as macro cells — pour-and-soak doesn’t reach into 5 µL chambers because of capillary forces.
Roughly $80–150 for macro, $100–180 for semi-micro, $140–220 for sub-micro, and $150–280 for ultra-micro. Premium sintered or molded fabrications add ~50% to those numbers. The cost gap is driven by the precision aperture machining for ultra-micro cells and the matched-set tolerance specs for premium spectroscopy work.
Most modern UV-Vis and fluorescence instruments accept the standard 12.5 × 12.5 × 45 mm body, so yes. The only caveat is Z dimension — masked ultra-micro cells must match the instrument’s beam height (15 mm modern vs 8.5 mm legacy). Open-chamber semi-micro cells are tolerant of small Z mismatches; masked ultra-micro cells are not.
Next step: confirm your Z dimension
Picking the right micro cuvette starts with two specs that must match your instrument: outer body envelope (almost always 12.5 × 12.5 × 45 mm) and Z dimension (15 mm modern, 8.5 mm legacy). Get those right and the cuvette class follows naturally from your sample volume.
The Z Dimension Lookup Tool covers 50+ instruments with confirmed Z values. If your instrument isn’t listed, send the model number with your custom-cell quote request — we’ll cross-reference and recommend the matching geometry.
📐 Find Your Instrument’s Z Dimension
Look up your spectrophotometer or fluorometer in seconds — 50+ models pre-cataloged · adapter spacers · custom-Z order path.
Open the Z Dimension Tool →



Recent Comments