Thermostatted Cuvettes for DNA Melting & Enzyme Kinetics
Thermostatted Cuvettes for DNA Melting & Enzyme Kinetics
Cuvette selection for temperature-controlled UV-Vis: which cell seals are evaporation-proof for 60-minute kinetic runs, which fit Peltier and water-jacketed holders, which work for DNA melting from 25 to 95 °C, and which let you do stopped-flow without losing alignment between shots.
Temperature is the second most important variable in a UV-Vis measurement after path length, and it is the variable that most often goes wrong silently. A 5 °C drift in sample temperature changes Beer-Lambert absorbance by 0.5–2 % for typical analytes due to refractive index, density, and molecular conformation effects. For routine endpoint measurements at room temperature this is negligible. For DNA melting curves where the entire experimental signal is a temperature scan, for enzyme kinetics where reaction rate doubles every 10 °C, or for thermal stability work where you are looking for sub-degree transitions, temperature control is everything — and the cuvette is part of the temperature control loop.
This guide covers what makes a cuvette suitable for temperature-controlled work: which seal types prevent evaporation during long kinetic runs, which formats fit Peltier and water-jacketed thermostatted holders, what wall thickness and material grade affect thermal response time, and which MachinedQuartz catalog categories are the right entry points for melting-curve, kinetic, and stopped-flow workflows. The decision matrix in section 8 maps eight common temperature-controlled experiment types to specific MQ product categories.
1. Why temperature matters in UV-Vis measurement
Three physical effects link sample temperature to apparent absorbance:
- Refractive index: water’s refractive index drops 0.0001 per °C; this changes Fresnel reflection at the cuvette window and shifts apparent absorbance by 0.05–0.1 % per °C.
- Sample density: water expands ~0.025 % per °C above 4 °C. Concentration in mol/L drops with temperature; the concentration in your cuvette at 50 °C is ~1 % less than the same sample at 20 °C.
- Molecular conformation: proteins and nucleic acids change conformation with temperature. A 1 °C shift can move a tryptophan A280 reading by 0.5 % through changes in tryptophan microenvironment.
Total effect for a typical biological sample: 0.5–2 % apparent absorbance change per 10 °C. For routine endpoint measurements where you are reading 0.5 AU and reporting to two significant figures, this is invisible. For methods that depend on temperature being a known constant — quantitative pharma assays, GMP IPC, NIST SRM-traceable work — you need the temperature locked. For methods that depend on temperature being a known variable — DNA melting, enzyme kinetics, thermal denaturation — you need the temperature scanned with calibrated stability.
2. The three temperature-control methods
Three distinct architectures cover essentially all UV-Vis temperature control. The cuvette choice is the same across all three.
Peltier (thermoelectric)
Solid-state thermoelectric cooler element in direct contact with the cuvette holder. Heats or cools by reversing current direction. Range 5–100 °C; stability ±0.1 °C. Standard accessory on Cary 3500, Lambda 365, Shimadzu UV-1900, JASCO V-770. No water plumbing required.
Water-jacketed
Hollow-walled holder with circulating water from external bath. Range −10 to +120 °C with appropriate fluid (water, ethylene glycol, silicone oil). Stability ±0.05 °C with quality circulator. Standard on legacy Cary, Lambda, and dedicated kinetic instruments.
Heated block
Resistive heating element embedded in the holder body. Range 25–200 °C; stability ±0.5 °C. Used in dedicated high-temperature instruments (OLIS, Aviv) and custom rigs. Asymmetric — can heat above ambient but cannot cool below it.
Choosing between them
If your spectrometer is from the past 10 years, it almost certainly comes with Peltier as a stock or low-cost accessory. Use it. If your work needs sub-zero temperatures, very high temperatures (above 100 °C), or you need ±0.05 °C stability for very precise melting curve work, a water-jacketed setup is the right choice. Heated blocks are reserved for instruments specifically designed for high-temperature absorbance — not a routine choice.
3. The three seal tiers — choose by experiment duration
Once you commit to a temperature different from ambient, the binding constraint becomes evaporation. A 10 °C temperature differential drives ~1 % volume loss per hour through an open cuvette top; a 30 °C differential (60 °C cuvette in a 30 °C lab) loses ~3 % per hour. The lost water concentrates the analyte and shifts measured absorbance by the same fraction. Three seal tiers handle different experiment durations.
Tier 1 — PTFE friction cap (open cuvette with cover)
The standard catalog cuvette ships with a PTFE press-fit cap. The cap reduces evaporation by about 60–70 % vs an open cell but does not seal hermetically. Suitable for: room-temperature endpoint measurements, kinetic runs under 10 minutes at modest temperature differentials (±15 °C from ambient), routine pharma QC where the method is well-defined.
Tier 2 — PTFE stopper (precision-fit ground stopper)
A PTFE-machined stopper fitted to a ground-glass receptacle on the cuvette body. Sealing performance roughly 95 % over 30 minutes; useful for kinetic runs of 10–60 minutes where a slight evaporation correction is acceptable. Easier to insert and remove than a screw cap, which matters when the protocol requires multiple sample additions during the experiment.
Tier 3 — screw-cap (sealed cuvette)
A threaded cap with a PTFE liner forms an essentially gas-tight seal. Sealing performance >99 % over multi-hour runs; mandatory for DNA melting (where the temperature scan duration is 30–90 minutes), enzyme kinetics longer than 60 minutes, anaerobic work, and any volatile-solvent application. Slight time penalty during loading because the cap must be threaded carefully without trapping bubbles.
For the deeper treatment of how each seal works, when to choose between them, and the cap-liner material trade-offs (PTFE / silicone / Viton / butyl / EPDM), see our screw-top cuvette guide.
 Tier 3 · sealed
Tier 3 · sealed
C102TR8 — 10 mm screw-cap
3.5 mL · two-way light · the workhorse for DNA melting and long enzyme kinetic runs
View screw-top range → Tier 2 · stoppered
Tier 2 · stoppered
C104SE6 — 10 mm stoppered fluor
3.5 mL · 4-clear-side · ground-stopper for fluorescence kinetics with reagent additions
View stoppered range →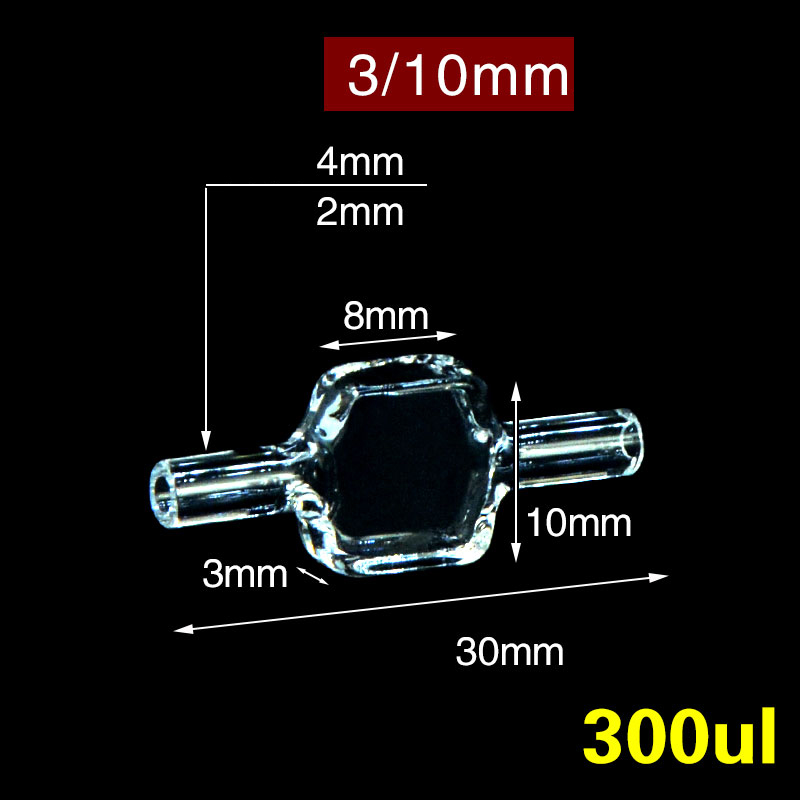 Stopped-flow
Stopped-flow
C034WS — 3 mm flow cuvette
300 µL · 4-way light · inlet/outlet ports for stopped-flow and continuous-flow kinetics
View flow cells →4. DNA / RNA / oligonucleotide melting curves
Thermal denaturation monitored at 260 nm is the textbook example of temperature-controlled UV-Vis. As double-stranded DNA melts to single strands, the unstacked bases lose their hypochromic shielding and absorbance increases by 30–40 %. The melting temperature Tm (midpoint of the sigmoid) is the diagnostic readout for primer design, mutation detection, and nucleic-acid characterisation.
Cuvette requirements for melting work
- Sealed cap mandatory. Scans run 30–90 minutes from 25–95 °C. An open or PTFE-friction-cap cell loses 3–8 % of volume to evaporation, concentrating the DNA and shifting the apparent A260 by the same factor. Use a screw-cap or precision-stoppered cell.
- Path length 10 mm for typical 50–500 ng/µL melting work. For very dilute samples (< 50 ng/µL), a 50 mm path or fluorescence-based detection (with intercalating dyes) makes more sense than a longer cell.
- Volume 200–500 µL in a 10 mm sub-micro or semi-micro cell minimises sample consumption. For 3.5 mL standard cells, the larger sample volume gives marginally better thermal stability but consumes more precious oligonucleotide.
- 4-clear-side useful for parallel A260 absorbance and fluorescence-based melting (intercalator dyes like SYBR Safe). 2-clear-side fine for absorbance-only work.
- JGS2 standard, JGS1 only if the application also touches deep UV below 230 nm.
Recommended scan parameters
- Heating rate: 0.5–1.0 °C/min for sharp Tm determination; up to 5 °C/min for screening.
- Temperature range: 25 to 95 °C covers most aqueous DNA work; for longer or more stable duplexes, extend to 99 °C.
- Mineral oil overlay is sometimes used as an additional evaporation barrier in non-sealed setups; sealed cuvettes make this unnecessary.
- Buffer: 10 mM Tris-HCl pH 7.5, 1 mM EDTA, 100 mM NaCl is the standard. Salt concentration shifts Tm by ~1 °C per 10 mM NaCl in the working range.
5. Enzyme kinetics — isothermal time-course measurements
Enzyme kinetics typically run isothermal at 25 °C (in vitro standard) or 37 °C (physiological). The temperature is held constant; the experimental signal is absorbance change over time as substrate is consumed or product accumulates. Kinetic runs span 30 seconds (fast enzymes, stopped-flow) to 60 minutes (slow enzymes or low substrate concentration); longer runs make sealing more important.
Common assays and cuvette implications
| Assay class | Typical λ | Run duration | Cuvette recommendation |
|---|---|---|---|
| NAD-dependent dehydrogenases (LDH, MDH, ADH, G6PDH) | 340 nm | 1–10 min | 10 mm with PTFE cap; sealed if > 5 min |
| Peroxidase / HRP coupled assays (TMB, ABTS) | 650 / 405 nm | 1–5 min | 10 mm with PTFE cap |
| Phosphatase (p-nitrophenyl phosphate) | 405 nm | 5–30 min | 10 mm stoppered or screw-cap |
| Protease (substrate-specific) | varies | 10–60 min | 10 mm screw-cap recommended |
| Long enzyme assays (slow turnover, low substrate) | varies | > 60 min | screw-cap mandatory |
| Cell-free transcription/translation | varies | 1–4 hours | screw-cap with septum (for sampling) |
The mid-experiment-addition problem
Many enzyme assays start with a buffer + substrate equilibration in the cuvette, then start the reaction by adding the enzyme. The cap must be removed for the addition, then replaced. Three options:
- PTFE friction cap: easiest to remove and replace. Acceptable for short assays where the temperature equilibration time after re-capping is short relative to the run.
- PTFE stopper: precision-fit, faster replacement than screw-cap; minimal temperature disturbance.
- Septum-cap (screw-cap with rubber septum): inject through the septum with a Hamilton syringe; cap never comes off. Best for sealed assays (anaerobic, volatile) but requires technique.
6. Stopped-flow kinetics — the flow-cell case
Stopped-flow spectroscopy measures very fast reactions (millisecond to second timescale) by mechanically combining two reagent streams in a small mixing chamber, then “stopping” the flow and recording the absorbance time course in the observation cell downstream of the mixer. The cuvette here is a flow cell with inlet and outlet ports, mounted in a temperature-controlled cell holder.
What stopped-flow needs from a cuvette
- Inlet and outlet ports (typically 4 mm OD glass-to-glass tubing connections) so the syringes can push reagent through the cell.
- Short path length — usually 1, 2, or 3 mm — to minimise dead time between the mixer and the observation point. Standard 10 mm paths are sometimes used for slow reactions but the dead time penalty is real.
- 4-clear-side or 2-clear-side depending on whether you also collect fluorescence (intrinsic fluorescence, bound dye, FRET).
- 200–700 µL volume typical for the observation chamber; smaller volumes give faster mixing but harder optical alignment.
- Standard 12.5 × 12.5 mm outer dimensions for compatibility with the manufacturer’s mounted cell block.
For the deeper treatment of flow cell geometry (square vs cylindrical, dead volume, mixing efficiency), see our flow cell category in the cuvette size chart.
7. Thermal response time and uniformity
Two physical properties determine how fast the sample inside the cuvette reaches the holder temperature: cuvette wall thickness and material thermal conductivity. Fused quartz has thermal conductivity 1.4 W/m·K, optical glass (BK7-class) about 1.1, PMMA only 0.19 (PMMA cuvettes do not work for fast temperature work). Wall thickness varies: thin-wall quartz (1 mm wall) equilibrates faster than standard (1.5 mm) than thick-wall glass (3 mm).
What this means for your experiment
- DNA melting at 0.5–1 °C/min: sample needs to track holder temperature within 0.1 °C. Thin-wall quartz; equilibration time is shorter than the scan step.
- Enzyme kinetic runs: after loading the buffer + substrate, allow 60–120 seconds for thermal equilibration before adding enzyme. Read-out during this window is not stable.
- Stopped-flow: the small flow-cell volume equilibrates to holder temperature in seconds; the binding constraint is that both syringe contents and the mixing-cell wall be pre-equilibrated, not the optical cell itself.
Temperature uniformity within the cuvette
A cuvette in a Peltier holder is heated/cooled from the bottom and sides through metal-to-quartz contact, and from the top via an open or capped air gap. The sample interior develops a small vertical thermal gradient (~0.1–0.3 °C) with the bottom warmer when heating, top warmer when cooling. The gradient is not visible in absorbance (the optical beam passes through the centre) but matters for very precise melting curve work where ±0.1 °C calibration is critical. Stirred cuvettes (with magnetic stir-bar or sonic agitation) eliminate the gradient at the cost of mechanical complexity.
8. Decision matrix — experiment to cuvette category
The matrix below maps eight common temperature-controlled UV-Vis experiment types to MachinedQuartz product categories. Use it as a starting point; specific path length, volume, and grade depends on your sample.
| Experiment | Recommended seal | Path | Geometry | MQ category |
|---|---|---|---|---|
| DNA melting (25→95 °C, 30–90 min) | Screw-cap (mandatory) | 10 mm | 2- or 4-side | Screw-Top Cuvettes |
| Protein thermal stability (CD or A280) | Screw-cap or precision stopper | 1–10 mm | 4-side preferred | Screw-Top or Stoppered |
| NAD-dependent kinetics, <5 min | PTFE friction cap acceptable | 10 mm | 2-side | Standard Cuvettes |
| Slow enzyme kinetics, 5–60 min | Stoppered or screw-cap | 10 mm | 2-side | Stoppered Cuvettes |
| Long enzyme kinetics, >60 min | Screw-cap with septum | 10 mm | 2-side | Screw-Top Cuvettes |
| Anaerobic / oxygen-sensitive kinetics | Screw-cap with septum | 10 mm | 2-side | Screw-Top Cuvettes |
| Stopped-flow kinetics (millisecond) | Flow cell with ports | 1–3 mm | 4-side | Flow Cells |
| Continuous-flow process monitoring | Flow cell with ports | 5–10 mm | 2-side | Flow Cells |
For DNA melting and any work above 60 °C, the screw-cap with PTFE liner is non-negotiable. For room-temperature kinetics under 5 minutes, the standard PTFE friction cap is fine. The middle range — 5 to 60 minute kinetic runs at 25 or 37 °C — is where the seal-tier choice has the biggest impact on data quality.
9. MachinedQuartz catalog entry points
Three product categories cover essentially all temperature-controlled cuvette work. Each is a stocked range with multiple SKUs across path length, volume, and geometry.
Need a thermostatted cuvette quote?
Send the experiment type (DNA melting / kinetics / stopped-flow), spectrometer model, and quantity. We respond within one business day with the right SKU and CoA documentation.
Request quote →Bulk & OEM →10. Related guides
11. Frequently asked questions
Yes, in the sense that the cuvette must be sealed for the duration of the temperature scan. DNA melting from 25 to 95 °C typically takes 30 to 90 minutes; an open or PTFE-friction-cap cuvette loses 3 to 8 percent of volume to evaporation during the run, concentrating the DNA and biasing the apparent A260 baseline. A screw-cap cuvette with PTFE liner is the standard choice. Outer dimensions, optical geometry (2-way or 4-way light), and path length (10 mm typical) are otherwise the same as for any other UV-Vis cuvette.
Yes. Peltier holders are designed to accept the universal 12.5 by 12.5 by 45 mm outer-dimension cuvette. The Peltier element makes thermal contact with the cuvette body via the holder’s metal walls. A standard MachinedQuartz 10 mm cuvette fits and works in any modern Peltier holder. The constraint is the cuvette seal, not the geometry: for runs over a few minutes at temperatures different from ambient, use a screw-cap or stoppered cuvette to prevent evaporation.
A stoppered cuvette has a PTFE or glass stopper that fits a ground-glass receptacle on the cuvette body — friction-fit, fast to insert and remove, sealing performance about 95 percent over 30 minutes. A screw-cap cuvette has a threaded cap with a PTFE liner — slower to insert because of threading, sealing performance over 99 percent over multi-hour runs, mandatory for DNA melting, anaerobic work, and any volatile-solvent application. Use stoppered when you need to make additions during the experiment; use screw-cap when you need a long, undisturbed sealed run.
Typical biological samples show 0.5 to 2 percent absorbance change per 10 degrees Celsius temperature change. The effects come from refractive-index changes (about 0.05 percent per degree), sample-volume expansion (water expands 0.025 percent per degree), and molecular conformation changes in proteins and nucleic acids. For routine endpoint measurements at known room temperature this is negligible. For pharmacopoeial methods, GMP IPC, and any quantitative work where two-significant-figure precision matters, lock the temperature with a Peltier or water-jacketed holder.
Yes, weakly. Fused quartz has thermal conductivity 1.4 W/m·K versus 1.1 for optical glass and 0.19 for PMMA. A thin-wall quartz cuvette (1 mm wall) equilibrates from 25 to 65 °C in about 30 seconds; standard 1.5 mm wall in about 90 seconds; thick-wall optical glass (3 mm wall) in about 4 minutes. For DNA melting at 0.5 °C per minute the equilibration time is short relative to the scan step. For fast kinetic work where the equilibration is the limiting step, choose thin-wall fused quartz.
No. PMMA softens at 80 to 100 °C and shows visible deformation by 70 °C; polystyrene by 90 °C. Both materials change dimensions when heated, biasing the optical path length. Polycarbonate is more thermally robust (Tg about 140 °C) but is rarely used for melting work because its UV transmission below 290 nm is poor and quartz is the validated material in published methods. Use fused quartz screw-cap cuvettes for DNA melting.
Modern Peltier accessory holders typically cover 5 to 100 °C with stability of plus or minus 0.1 °C. Some specialty Peltier modules extend to minus 10 °C with active cooling, or up to 110 °C with high-current configurations. Beyond this range, water-jacketed holders (minus 10 to plus 120 °C with appropriate fluid) or heated-block holders (25 to 200 °C) are the right choice. Check your spectrometer vendor’s accessory specifications.
For most enzyme kinetics at room temperature with well-mixed reagents, no. The cuvette volume (3.5 mL standard) is small enough that diffusion completes mixing in 1 to 2 minutes. For reactions where mixing is the rate-limiting step (very fast enzymes, viscous samples, particulate suspensions), a magnetic stir-bar in the cuvette plus a stirrer accessory in the cell holder eliminates the diffusion delay. Some screw-top cuvettes are designed to accept a small stir bar; check before ordering if stirring is required.
No. NanoDrop pedestals are single-point measurement instruments — pipette, measure, wipe. They do not support continuous-monitoring kinetic mode and do not have temperature control suitable for kinetic work. For enzyme kinetics, use a UV-Vis benchtop spectrophotometer with a Peltier or water-jacketed cell holder and a sealed cuvette. See our cuvette-versus-NanoDrop guide for the comparison.
Typical Peltier modules ramp at 1 to 5 °C per minute. Reaching plus or minus 0.5 °C of a new setpoint takes 5 to 30 minutes depending on the temperature delta. Once at setpoint, stability is plus or minus 0.1 °C indefinitely. For DNA melting workflows, scan rate options of 0.1 to 5 °C per minute are typical; 0.5 to 1.0 °C per minute is the sweet spot for sharp Tm determination. Faster scans give Tm estimates with about 1 to 2 °C systematic error toward higher temperatures (kinetic lag).
12. Disclaimer & notes
Recommendations on this page are general guidance for typical UV-Vis temperature-controlled work. Specific assays, regulatory environments, or instrument-vendor recommendations may shift the choice. For pharmacopoeial methods (USP, EP, JP) follow the cuvette specification in the published method.
Temperature ranges and stability values for Peltier and water-jacketed holders are typical of modern instruments at the time of writing. Verify with your spectrophotometer vendor’s accessory specifications for binding figures.
Water-jacketed cuvettes vs holders. MachinedQuartz manufactures standard cuvettes that fit water-jacketed cell holders. We do not manufacture integrated water-jacketed cuvettes (cuvettes with welded outer water jackets) — for that specialty form factor, consult Hellma 121 series or equivalent. The Peltier-plus-standard-cuvette workflow described in this guide is the dominant modern approach and covers 95 percent of temperature-controlled UV-Vis work.
Trademark notice: Cary, Lambda, Shimadzu, JASCO, OLIS, Aviv, Hellma, Hamilton are trademarks of their respective owners. References are for compatibility and method context only.
Information currency: last reviewed May 2026.




Recent Comments